This is a rare disorder characterised by tendency to severe peptic ulceration, that occurs secondary to hypersecretion of gastric acid due to unregulated gastrin release from a non-beta cell, often well-differentiated neuroendocrine tumor (NET, gastrinoma).
It presents aggressive and refractory ulceration in which total gastrectomy provided the only chance for enhancing survival.
Pathophysiology
-
The main etiologic factor is hypergastrinemia originating from an autonomous neoplasm. These tumors are commonly found (60-90%) within the hypothetical gastrinoma triangle, formed by the confluence of,
- Cystic and common bile ducts superiorly,
- Junction of the second and third portions of the duodenum inferiorly, and
- Junction of the neck and body of the pancreas medially.
-
The increased gastrin production leads to increased gastric acid production through,
- Parietal cell stimulation by gastrin receptors.
- Increased parietal cell mass.
- Induction of histamine release from ECL cells.
-
Gastrin also have a trophic acid on gastric epithelial cells.
-
The increased gastric acid output leads to,
- Peptic ulcer diathesis,
- Erosive oesophagitis, and
- Diarrhoea.
-
Etiology of diarrhoea is multifactorial and it may result due to,
- Marked volume overload to the small bowel.
- Pancreatic enzyme inactivation by acid.
- Damage to the intestinal surface by acid.
- Direct stimulatory effect of gastrin on enterocytes or the co-secretion of additional hormones from the tumor such as vasoactive intestinal peptide.
Clinical Features
-
Abdominal pain in the presence of acid peptic disorders (peptic ulcer).
-
Multiple, severe peptic ulcers may present in unusual sites such as the post-bulbar duodenum, jejunum or oesophagus.
-
Symptoms of oesophageal origin ranging from mild oesophagitis to frank with stricture and Barrett's mucosa.
-
Diarrhoea (in 70% of patients).
-
ZES patients with MEN 1 syndrome (found in ~25% of patients)
- Hyperparathyroidism and hypercalcemia seen in MEN 1 patients may have a direct effect on ulcer disease (due to the stimulatory effect of calcium on gastric secretion).
- A reduction in gastrin and gastric acid output is seen after resolution of hypercalcemia by parathyroidectomy.
- Have higher incidence of gastric carcinoid tumor development.
- Have a more indolent course as compared to patients with sporadic gastrinomas.
- Gastrinomas tend to be smaller, multiple and located in the duodenal wall more often.
Investigations
It involves biochemical tests to confirm the diagnosis, followed by an abdominal CT scan, MRI or OctreoScan/PET-CT with Ga-DOTATATE to exclude metastatic disease.
Once metastatic disease has been excluded, one may opt for exploratory laparotomy with intra-operative ultrasound or transillumination (where experienced endocrine surgeon available) or careful examination of the peri-pancreatic area with EUS, accompanied by endoscopic exploration of the duodenum for primary tumors.
Fasting gastrin levels
-
Normal : Less than 150 pg/ml.
-
Gastrinoma patients will have a gastrin level > 150-200 pg/ml. Fasting gastrin more than 10 times normal is highly suggestive of ZES.
-
Use of PPI makes the diagnosis somewhat difficult, as they lower acid secretion and elevate fasting gastrin levels in normal individuals.
-
Measurement of fasting gastrin should be repeated to confirm the clinical suspicion. A normal fasting gastrin on two separate occasions, especially if the patient is on a PPI, virtually excludes this diagnosis.
-
PPI should be stopped for a minimum of 7 days before testing and the patient be placed on a histamine H2 antagonist such as famotidine (2-3 times per day). H2 antagonist should be stopped 24 hr before. Patient may take antacids for the final day, stopping them around 12 hr before testing.
-
Elevated fasting gastrin levels can be found in :
- Patients using anti-secretory agents.
- H. pylori infection.
- Retained gastric antrum.
- G-cell hyperplasia.
- Gastric outlet obstruction.
- Renal insufficiency.
- Massive small-bowel obstruction.
- Conditions such as rheumatoid arthritis, vitiligo, diabetes mellitus and pheochromocytoma.
Assessment of acid secretion
-
Decreased acid output in the absence of a PPI is highly suggestive and nothing further needs to be done.
-
pH can be measured in gastric fluid obtained either during endoscopy or through nasogastric aspiration,
- pH < 3 is suggestive of a gastrinoma.
- pH > 3 : Formal gastric acid analysis should be performed.
- Normal BAO in nasogastric surgery patients is typically < 5 meq/h. BAO < 5 meq/h in the presence of hypergastrinemia is pathognomonic of ZES.
- BAO/MAO ratio using pentagastrin infusion to maximally stimulate acid production : More than 0.6 highly suggestive of ZES.
Gastrin provocative tests
- Developed to differentiate between the causes of hypergastrinemia, especially helpful in patients with intermediate acid secretory studies.
- Secretin stimulation study : Most sensitive and specific gastrin provocative test for the diagnosis of gastrinoma. An increase in gastrin levels to or above 120 pg within 15 min of secretin injection has a sensitivity and specificity of more than 90% for ZES.
- Calcium infusion study : Rarely used due to cumbersome nature of the test and lower sensitivity and specificity than secretin stimulation.
- PPI must be stopped for 1 week before testing (PPI-induced hypochlorhydria or achlorhydria may lead to a false-positive secretin test).
Tumor localisation
-
Endoscopic ultrasound (EUS)
- Permits imaging of the pancreas with a high degree of resolution (< 5mm).
- Helpful in excluding small neoplasms within the pancreas and in assessing the presence of surrounding lymph nodes and vascular involvement.
- Not very sensitive (43%) for finding duodenal lesions.
-
Radio-labelled somatostatin receptor scintigraphy (Cell surface receptors for somatostatin)
-
Permits the localisation, staging and prediction of therapeutic response to somatostatin analogues by gastrinomas.
-
The scintigraphic tools developed for measuring the uptake of the stable somatostatin analogue includes,
- In-pentetreotide (OctreoScan) : Sensitivity and specificity > 80%.
- PET-CT with Ga-DOTATATE : Sensitivity and specificity > 90%.
- F-Fluorodeoxygenase (F-FDG) PET imaging
-
-
Selective arterial secretin injection may be useful adjuvant for localising tumors in a subset of patients.
Management
Management of functional endocrine tumors is directed at
- reducing the signs and symptoms related to hormone overproduction,
- curative resection of the neoplasm and
- attempts to control tumor growth in metastatic disease.
Symptomatic treatment
-
PPIs are the treatment of choice.
- Initial dose (Omeprazole, Lansoprazole, Rabeprazole or Esomeprazole) : In the range of 60 mg in divided doses in a 24 h period.
- It is important to closely monitor the clinical symptoms when starting PPIs and increase the dose accordingly.
- PPIs have reduced the need for total gastrectomy and in the majority of patients continuous therapy with omeprazole heals ulcer and alleviates diarrhoea.
-
Somatostatin analogues : Octreotide or Lantreotide may be considered as adjunctive therapy to PPI in patients with tumors that express somatostatin receptors and have peptic symptoms that are difficult to control with high-dose PPI.
Surgical treatment
- The goal is to provide a definitive cure.
- Approximately 30% of small and single tumors can be localised and resected but many tumors are multifocal.
- Some patients present with metastatic disease and surgery is inappropriate.
Treatment for metastatic endocrine tumors
- Temozolomide with capecitabine : Demonstrated radiographic regression (70%) and progression-free survival (18 months) in patients with well-differentiated NETs.
- Systemic therapy with radiolabeled somatostatin analogues (peptide receptor radiotherapy, PPRT).
Prognosis
The overall 5-year survival rate is 62-75% and for 10-year 47-53%.
Favourable prognostic indicators include:
- Primary duodenal wall tumors.
- Isolated lymph node tumor.
- Presence of MEN 1.
- Undetectable tumor upon surgical exploration.
Poor outcome is seen in:
- Patients with shorter disease duration.
- Female sex.
- Older age at diagnosis.
- Higher gastrin levels (> 10,000 pg/ml).
- Poor histologic differentiation.
- High proliferative index.
- Large pancreatic primary tumors (> 3 cm).
- Metastatic disease to lymph nodes, liver and bone.
- Cushing's syndrome.
- Rapid growth of hepatic metastases.
Points to Note
- There is decreased referral for gastrinoma, with delay in diagnosis and increased false-positive diagnoses of ZES, due to widespread availability and use of PPIs.
- Most common non-pancreatic lesion (gastrinoma) : Duodenal tumors (60-100%)
- The acid output may be so great that it reaches the upper small intestine, reducing the luminal pH to 2 or less. This can damage the epithelial cells and lead to a mild degree of maldigestion and malabsorption of nutrients.
- MEN 1 syndrome is caused by inactivating mutations of MEN1 tumor-suppressor gene found on the long arm of chromosome 11q13 and primarily involves the parathyroid glands (80-90%), pancreas (40-80%) and pituitary glands (30-60%).
- Gastrinoma patients should be screened for MEN 1 by performing a detailed family history and obtaining several serum markers including calcium, parathyroid, prolactin and pancreatic polypeptide levels.
- The diagnosis of MEN 1 is critical in order to minimise the long-term sequelae of gastric acid hypersecretion, prevent metastatic disease, provide genetic counselling to the patient and his or her family and also to determine the recommended surgical approach.
References
-
Harrison's Principles of Internal Medicine (21st edition), Joseph Loscalzo & Anthony S. Fauci & Dennis L. Kasper & Stephen Hauser & Dan Longo & J. Larry Jameson, The McGraw-Hill Companies.
-
The image used is licensed under the Creative Commons Attribution-Share Alike 3.0 Unported license.
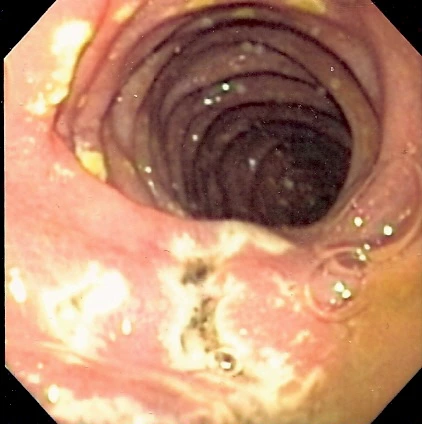
- Description : Endoscopic image of multiple small ulcers located in the distal duodenum in a patient with gastrinoma (Zollinger-Ellison syndrome).
- Source : http://en.wikipedia.org/wiki/Image:ZES_endo.jpg
- Author : http://en.wikipedia.org/wiki/User:Samir_%28The_Scope%29
*This article is an excerpt from the above mentioned book and Medical Sutras does not make any ownership or affiliation claims.