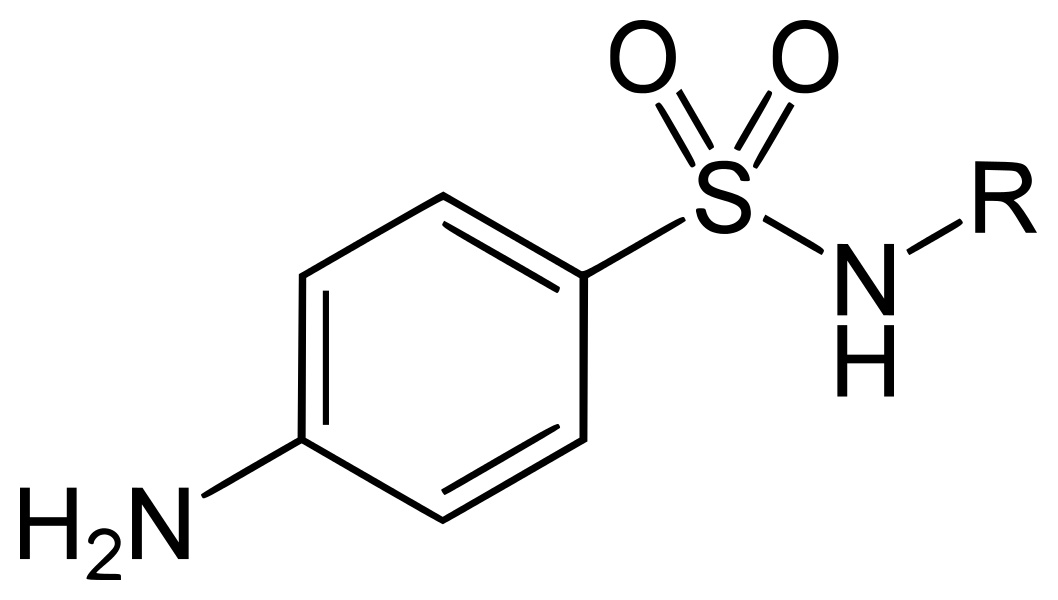
The sulfonamide group of drugs are derivatives of sulfanilamide (p-aminobenzene sulfonamide). The sulfur is linked directly to the benzene ring, and the para-NH2 group (the N of which is designated as N4) can be replaced by moieties that can be converted in vivo to a free amino group.
They were the first antimicrobial agents effective against pyogenic bacterial infections. Clinically relevant sulfonamides include,
- Short acting : Sulfadiazine.
- Intermediate acting : Sulfamethoxazole.
- Long acting : Sulfadoxine, Sulfamethopyrazine.
- Special purpose : Sulfacetamide sodium, Sulfasalazine, Silver sulfadiazine, Mafenide.
Antibacterial Spectrum
Sulfonamides are primarily bacteriostatic agents, effective against both Gram-positive and Gram-negative bacteria. Although, several microbes have become resistant, those still sensitive include:
- Streptococcus pyogenes, Haemophilus influenzae, H. ducreyi, Calymmatobacterium granulomatis, Vibrio cholerae.
- Chlamydia, Actinomyces, Nocardia and Toxoplasma.
- Anaerobic bacteria are not susceptible.
Mechanism of Action
Sulfonamides being structural analogues of p-aminobenzoic acid (PABA), inhibit bacterial folate synthesis.
- Several bacterias synthesise their own folic acid of which PABA is a constituent (taken up from the medium).
- Sulfonamides competitively inhibit dihydropteroate synthase, the enzyme responsible for the incorporation of PABA into dihydropteroic acid (immediate precursor of folic acid).
Pteridine + PABA -> Dihydropteroic acid
Dihydropteroic acid + Glutamate -> Dihydrofolic acid
- Also, sulfonamides, being chemically similar to PABA, may itself get incorporated to form an altered folate which is metabolically injurious.
Bacterial resistance may occur due to:
- Production of increased amount of PABA.
- Decrease in affinity of folate synthase enzyme for sulfonamides.
- Adoption of alternate pathway for folate metabolism.
Pharmacokinetics
Absorption : Rapidly and nearly completely absorbed from the g.i. tract. Peak plasma levels are achieved in 2-6 hours, depending on the drug.
Distribution
- Widely distributed in the body, readily enters body fluids and cross placenta freely.
- The free form of sulfadiazine attains similar concentration in CSF as in plasma.
- The extent of plasma protein binding differs considerably (10-95%) among different sulfonamides, depending on the hydrophobicity of the drug and its pKa.
- Highly protein bound sulfonamides are longer acting.
Metabolism
- Primarily occurs in the liver by acetylation at N4 by non-microsomal acetyl transferase.
- The acetylated derivative is inactive, but may contribute to the adverse effects. It is generally less soluble in acidic urine than the parent drug and may precipitate causing crystalluria.
Excretion : Mainly by the kidney through glomerular filtration.
Adverse Effects
All sulfonamides show relatively common side effects.
- Nausea, vomiting and epigastric pain.
- Crystalluria : Dose related (less frequent now). It can be minimised by taking plenty of fluids and by alkalinising the urine.
- Hypersensitivity reactions (2-5% patients) : Stevens-Johnson syndrome and exfoliative dermatitis are serious reactions reported with the long-acting agents.
- Hepatitis (0.1% patients).
- Hemolysis : Can occur in G6PD deficiency with high doses of sulfonamides.
- Kernicterus : May be precipitated in newborns (esp. premature).
Indications
Sulfonamides are used in combination with other drugs.
- Trimethoprim + Sulfamethoxazole (Cotrimoxazole) : Used for many bacterial infections, Pneumocystis jiroveci and nocardiosis.
- Sulfadoxine, Sulfamethoxypyrazine with pyrimethamine : Used for malaria, Pneumocystis jiroveci pneumonia in AIDS patients and toxoplasmosis.
- Silver sulfadiazine or Mafenide : Used topically (1% cream) for preventing infection on burn surfaces.
- Sulfacetamide sod. (10-30%) : Used topically for ocular infections due to susceptible bacteria and chlamydia, including ophthalmia neonatorum caused by Ch. oculogenitalis.
Points to Note
- Sulfonamides are active against only those microbes which cannot take folic acid from outside and have to synthesise their own folic acid. Hence, they do not interfere with host cells (utilise preformed folic acid supplied in diet).
- PABA, in small quantities, antagonises the antibacterial action of sulfonamides.
- Pus and tissue extracts antagonise sulfonamide action as they contain purines and thymidine which decreases bacterial requirement for folic acid. Pus is also rich in PABA.
References
- Laurence L. Brunton - Goodman & Gilman's Manual of Pharmacology and Therapeutics-McGraw-Hill Medical (2008).
- Essentials of Medical Pharmacology (8th Edition), K.D. Tripathi -Jaypee Brothers Medical Publishers (P) Ltd.
- Basic and Clinical Pharmacology (15th Edition), Bertram G. Katzung, Todd W. Vanderah, McGraw Hill Lange.
*This article is an excerpt from the above mentioned books and Medical Sutras does not make any ownership or affiliation claims.