The cellular response to injury depends on:
- The type of injury, its duration and severity, and
- The type, status, adaptability and genetic makeup of the injured cell.
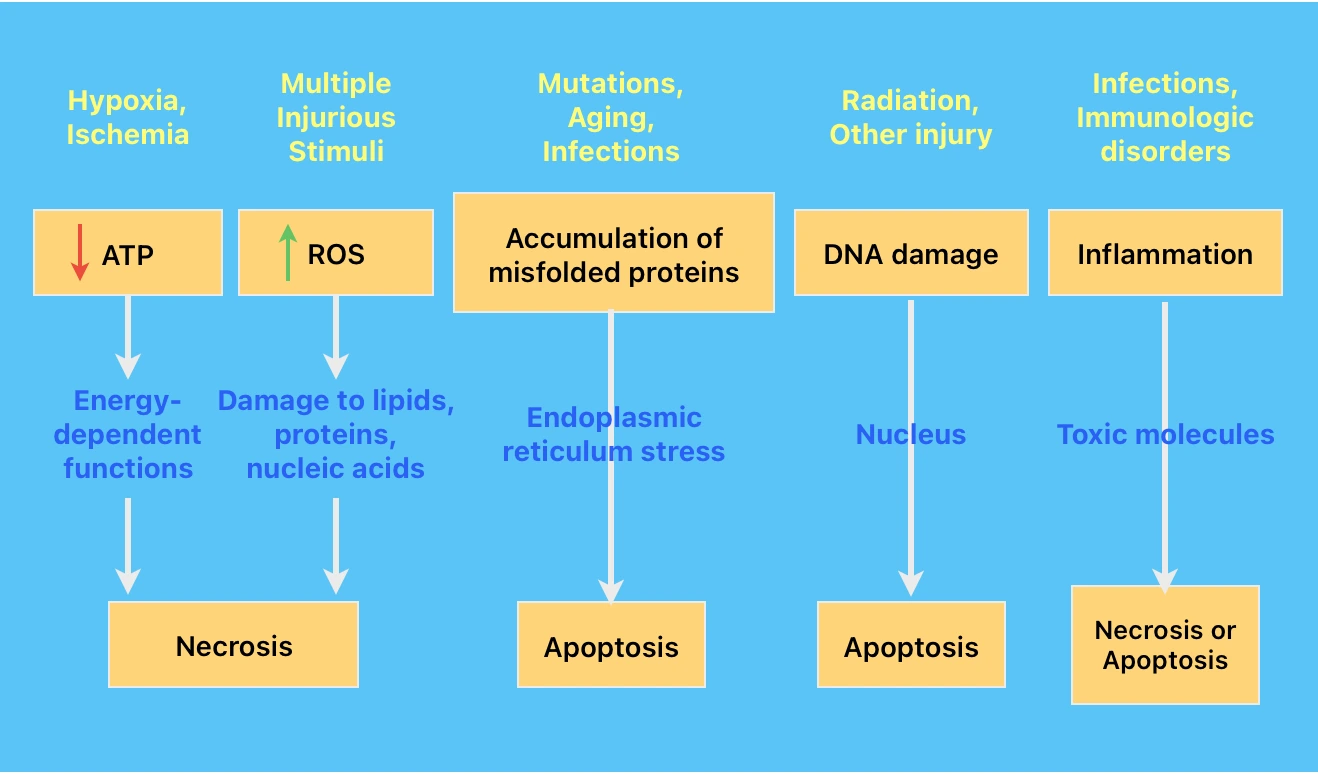
The principal biochemical mechanisms involved in cell injury and death can be described based on the type of injury and some abnormalities that characterise cell injuries regardless of the cause.
Mechanisms based on etiology
- Failure of energy-dependent metabolic pathways: Hypoxia & Ischemia.
- Increase in Reactive Oxygen Species (ROS)/Oxidative stress: Multiple injurious stimuli.
- Toxins.
- Accumulation of misfolded proteins (Endoplasmic reticulum stress): Mutation, cell stress, infections.
- Inflammation: Infections & immunologic disorders.
- DNA damage: Radiation/chemotherapeutic agents, intracellular ROS, mutations.
Mechanisms associated with diverse causes
- Mitochondrial dysfunction.
- Defects in membrane permeability.
Hypoxia & Ischemia
Hypoxia (oxygen deficiency) and ischemia (reduced blood supply), both deprive tissues of oxygen and oxygen deprivation is one of the most common causes of cell injury and necrosis.
Pathophysiology
-
Cells when subjected to low oxygen levels initially activate compensatory mechanisms induced by transcription factors of hypoxia-inducible factor 1 (HIF-1) family.
-
HIF-1 stimulates synthesis of several proteins that help the cell survive low oxygen levels. These include:
- Vascular endothelial growth factor (VEGF): Stimulates growth of new blood vessels and thus help in increasing blood flow and oxygen supply.
- Other proteins stimulate the uptake of glucose and glycolysis, thus dampening mitochondrial oxidative phosphorylation. Anaerobic glycolysis can generate ATP in the absence of oxygen using glucose either from the circulation or from the hydrolysis of intracellular glycogen.
-
However, persistent or severe hypoxia and ischemia ultimately lead to failure of ATP generation and depletion of ATP (energy stores) in cells. (Most cellular ATP is produced from ADP by oxidative phosphorylation during reduction of oxygen in the electron transport system of mitochondria.)
Effects of ATP Depletion
- Reduced activity of ATP-dependent plasma membrane sodium pumps: Leads to intracellular accumulation of sodium and efflux of potassium i.e., a net gain of solute. This is accompanied by isosmotic gain of water, resulting in cell swelling and dilation of the ER.
- Increase in anaerobic glycolysis: Leads to lactic acid accumulation, decreased intracellular pH, and decreased activity of many cellular enzymes.
- Structural disruption of protein synthetic apparatus: There is disruption of ribosomes from the rough ER and dissociation of polysomes into monosomes, with consequent reduction in protein synthesis.
- Irreversible damage to mitochondrial and lysosomal membranes.
Ischemia-Reperfusion Injury: Under certain circumstances, the restoration of blood flow to ischemic but viable cells, results in increased cell injury. This may result from:
- Increased generation of ROS.
- Additional tissue injury by activated leukocytes that comes with reperfusion.
Reactive Oxygen Species
Reactive oxygen species belong to a group of molecules (free radicals) that consist of a single unpaired electron in outer orbit e.g., Superoxide, Hydrogen peroxide, Hydroxyl radical, Peroxynitrite.
These are extremely unstable, and readily react with inorganic and organic molecules. The resulting cellular abnormalities are referred as oxidative stress.
Pathophysiology
Cell injury results from the excessive accumulation of ROS, which is determined by their rates of production and removal.
Production of ROS
- Normally in small amounts in all cells during reduction-oxidation(redox) reactions that occur during mitochondrial respiration and energy generation.
- In phagocytic leukocytes, mainly neutrophils and macrophages, for destruction of ingested microbes and other substances during inflammation and host defense.
Removal of ROS
- Inherently unstable and decay spontaneously.
- Free radical scavengers: Superoxide dismutase, Glutathione peroxidase, Catalase, endogenous/exogenous antioxidants such as vitamin E, A and C and beta-carotene.
Effects of Accumulated ROS
- Lipid peroxidation of membranes: The polyunsaturated lipids in membrane react with free radical and forms peroxides, which are themselves unstable and reactive. This leads to an autocatalytic chain reaction damaging the plasma membranes and mitochondrial and lysosomal membranes.
- Crosslinking and other changes in proteins: Free radicals promote sulfhydryl-mediated protein crosslinking, resulting in enhanced degradation or loss of enzymatic activity.
- DNA damage: Free radicals react with thymine residues in nuclear and mitochondrial DNA and produces single-strand breaks.
Toxins
Environmental chemicals and substances produced by infectious agents causes cell injury and primarily necrosis by two general mechanisms:
Direct-acting Toxins: These act directly by combining with a critical molecular component or cellular organelle. For example,
- In mercuric chloride poisoning, mercury binds to the sulfhydryl groups of various cell membrane proteins and cause inhibition of ATP-dependent transport and increased membrane permeability.
- Many antineoplastic chemotherapeutic agents induce cell damage by direct cytotoxic effects.
- Toxins made by microorganisms often cause damage by targeting host cell molecules that are required for essential functions, such as protein synthesis and ion transport.
Latent Toxins: These include toxins that are not intrinsically active but damage target cells when converted to reactive metabolites. They typically affect the cells in which they are activated and this is usually accomplished by cytochrome P450 in the smooth ER of liver and other organs.
The common mechanisms include:
- Direct covalent binding to protein and lipids, resulting in membrane damage and cell injury.
- Formation of free radicals such as in case of carbon tetrachloride (used in the dry cleaning industry) and the analgesic acetaminophen.
Accumulation of Misfolded Proteins (Endoplasmic Reticulum Stress)
The intracellular accumulation of misfolded proteins can be caused by several factors that increase their production or reduce the ability to eliminate them. These include:
- Gene mutations, leading to production of proteins that cannot fold properly.
- Aging, associated with decreased capacity to correct misfolding.
- Infections (esp. viral), leads to synthesis of large amount of microbial proteins than the cells can handle.
- Increased demand of secretory proteins such as insulin in insulin-resistant states.
- Changes in intracellular pH and redox state.
Pathophysiology
- Normal protein synthesis: Chaperons in ER control proper folding of newly synthesised proteins and misfolded polypeptides are ubiquitinated and targeted for proteolysis.
- Unfolded protein response: Protective cellular response in response to accumulation of unfolded or misfolded proteins in the ER. This is the initial response that activates signaling pathways which leads to increased production of chaperones and decrease protein translation, thus reducing the levels of misfolded proteins in the cell.
- Apoptosis: This occurs when large amount of misfolded protein accumulates, that cannot be handled by the adaptive response. There is activation of proapoptotic sensors of the BH3-only family and direct activation of caspases, which leads to apoptosis by the mitochondrial (intrinsic) pathway.
Diseases caused by misfolded proteins
-
Protein degradation and deficiency
- Cystic fibrosis (Cystic fibrosis transmembrane conductance regulator).
- Familial hypercholesterolemia (LDL receptor).
- Tay-Sachs disease (Hexosaminidase beta subunit).
-
ER stress-induced cell loss
- Retinitis pigmentosa (Rhodopsin).
- Creutzfeldt-Jacob disease (Prions).
- Alzheimer disease (A-beta peptide).
-
Both ER stress-induced cell loss and functional protein deficiency
- Alpha-I-antitrypsin deficiency (alpha-I antitrypsin).
References
- Robbins Basic Pathology, 10th edition, Vinay Kumar, Abul K. Abbas, Jon C. Aster, Elsevier.
*This article is an excerpt from the above mentioned book and Medical Sutras does not make any ownership or affiliation claims.