Helicobacter pylori initially named Campylobacter pyloridis is a gram-negative micro-aerophilic rod (S-shaped, with multiple sheathed flagella) found most commonly in the deeper portions of the mucous gel coating the gastric mucosa or between the mucous layer and the gastric epithelium.
The bacteria is prevalent throughout the world (depends to a great extent on the overall standard of living) : In developing countries, 80% of the population may be infected by the age of 20, while in developed countries, the prevalence is 20-50%.
Predisposing / Risk factors
- Poor socio-economic status.
- Low literacy rate.
- Birth or residence in a developing country.
- Domestic crowding.
- Unsanitary living conditions.
- Unclean food or water.
- Exposure to gastric contents of an infected individual : Transmission of H. pylori occurs from person to person, following an oral-oral or fecal-oral route.
Pathophysiology
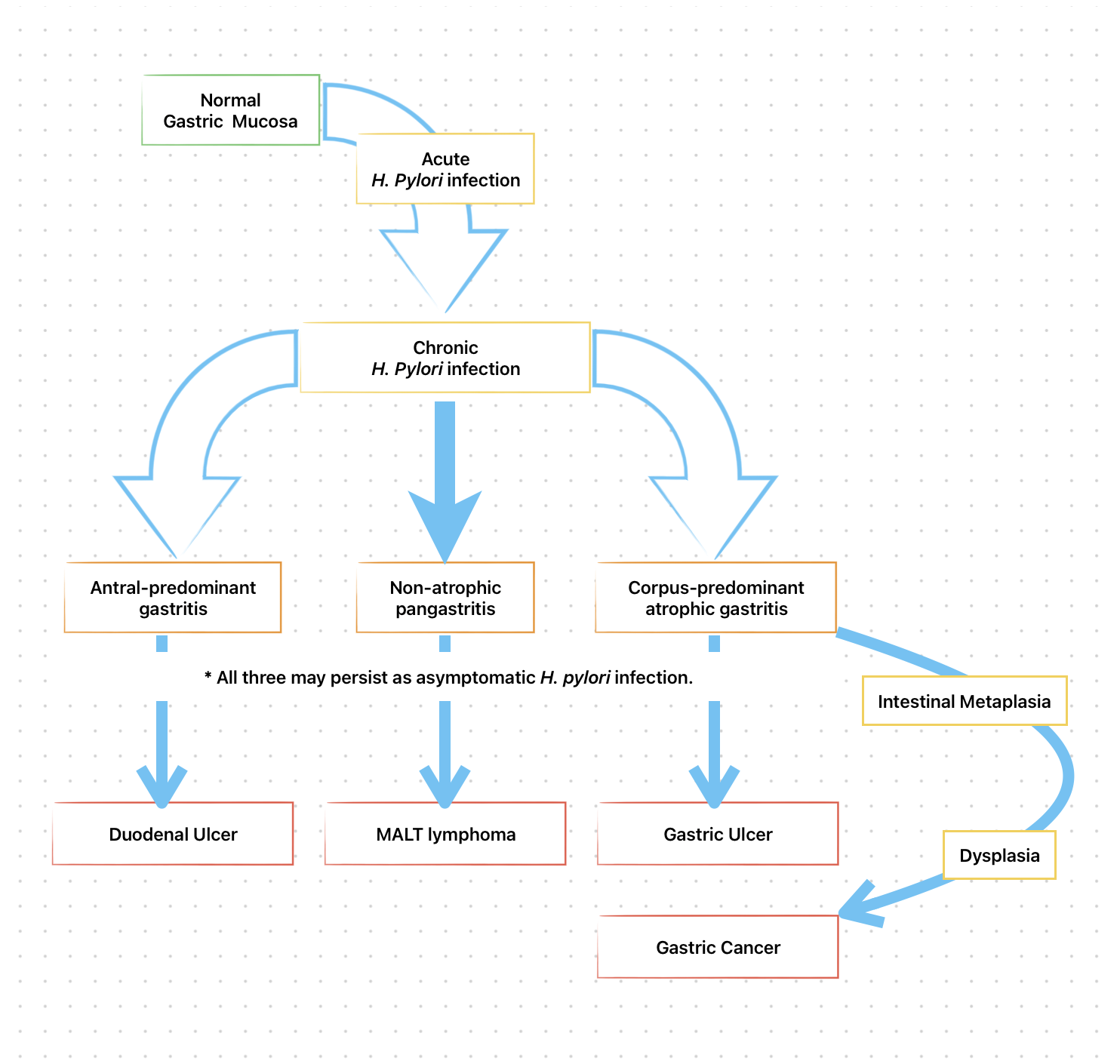
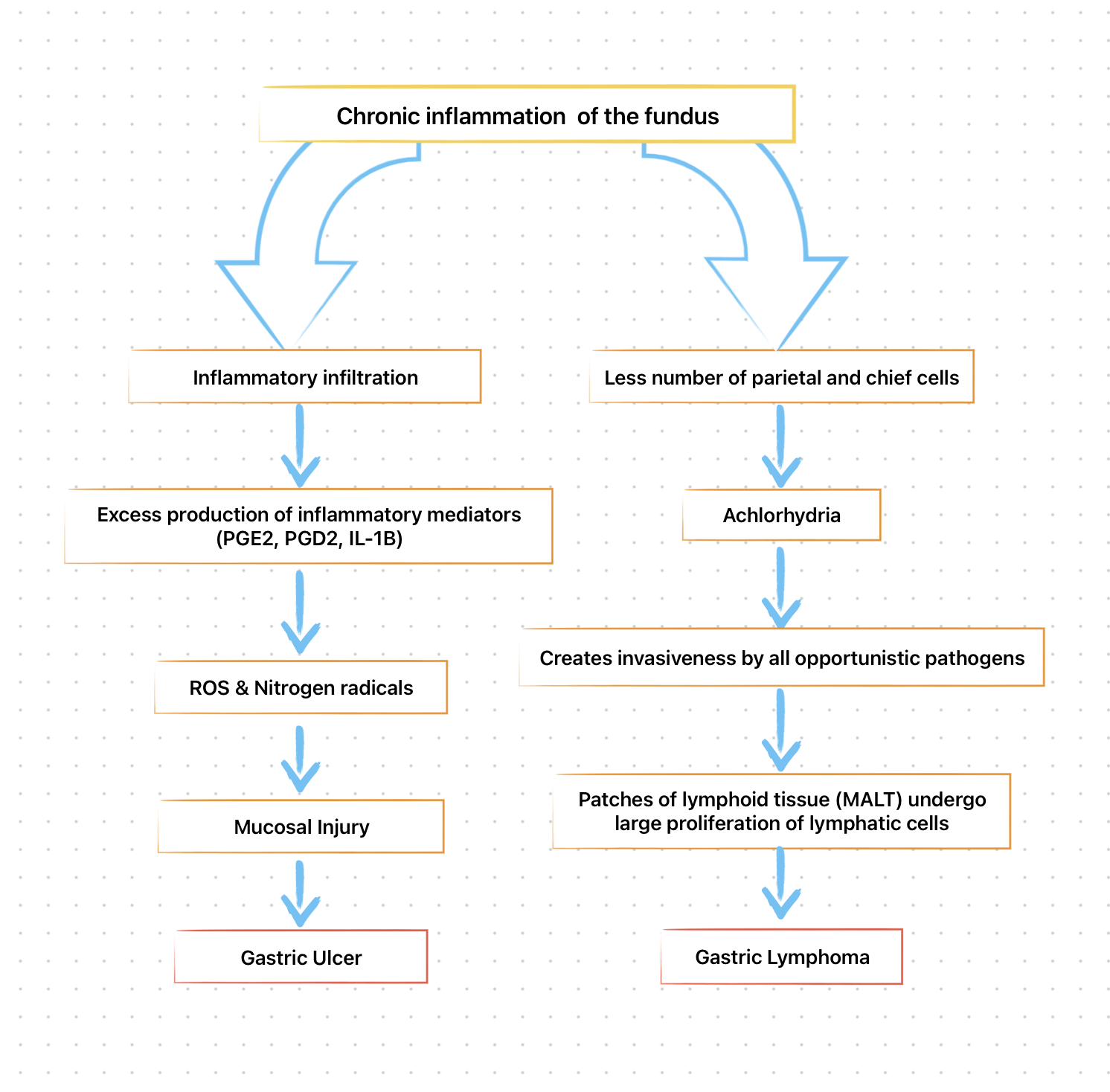
- H pylori infection is virtually always associated with a chronic active gastritis, but 10-15% of infected individuals develop frank peptic ulceration.
- The bacterium is present in only 30-60% of individuals with gastric ulcers and 50-70% of patients with duodenal ulcers.
- The particular end result of H. pylori infection (gastritis, PUD, gastric MALT lymphoma, gastric cancer) is determined by a complex interplay between bacterial and host factors.
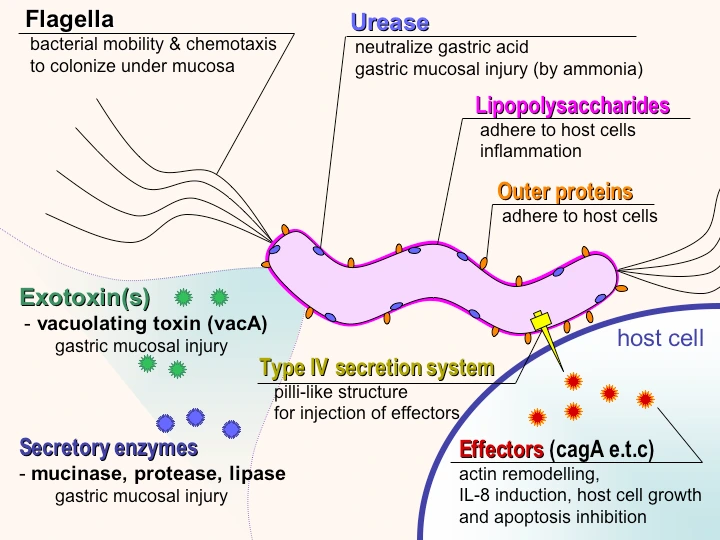
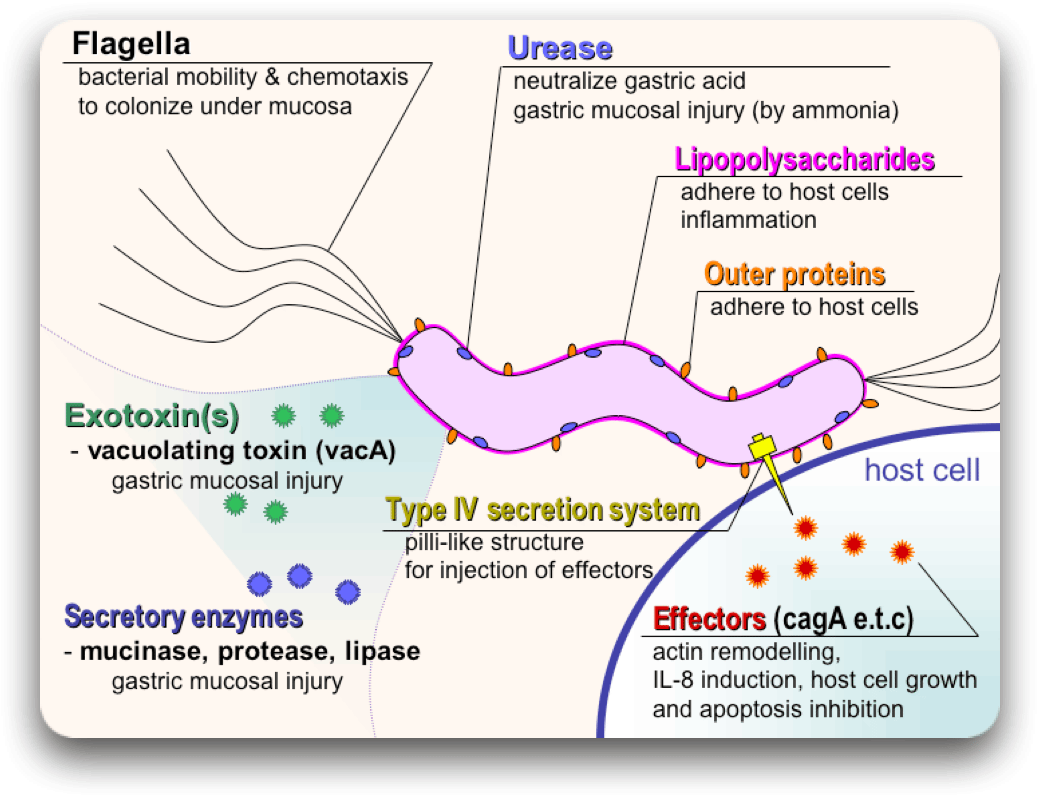
Bacterial factors
Different strains of H. pylori produce different virulence factors that in conjunction with additional bacterial constituents helps in gastric residence, induce mucosal injury and avoid host defense.
- Structure : The organism is capable of transforming into a coccoid form, which represents a dormant state that may facilitate survival in adverse conditions.
- Cytotoxin associated gene pathogenicity island (Cag-PAI) : Specific region of the bacterial genome that encodes virulence factors Cag A and pic B. Cag-A-dependent mechanism may directly inhibit parietal cell H+,K+-ATPase activity leading to low acid production.
- Vacuolating cytotoxin (VacA) targets human CD4T cells, inhibiting their proliferation and can disrupt normal function of B cells, CD8 T cells, macrophages and mast cells.
- Urease allows H. pylori to reside in the acidic stomach and generates ammonia from urea (alkalinise surrounding pH), which can damage epithelial cells.
- Surface factors : Are chemotactic for neutrophils and monocytes, which in turn contribute to epithelial cell injury.
- Proteases and phospholipases : Break down the glycoprotein lipid complex of the mucosal gel, thus reducing the efficacy of this first line of mucosal defense.
- Adhesins (Outer membrane proteins like BabA): Facilitate attachment of the bacteria to gastric epithelial cells.
- Lipopolysaccharide (LPS) : Has low immunologic activity compared to that of other organisms, but may promote a smoldering chronic inflammation.
 *This file is licensed under the Creative Commons Attribution-Share Alike 3.0 Unported license. Source : Wikimedia Commons (Y_tambe's file), Author : user:Y_tambe, Permission : GFDL, CC-by-sa.
*This file is licensed under the Creative Commons Attribution-Share Alike 3.0 Unported license. Source : Wikimedia Commons (Y_tambe's file), Author : user:Y_tambe, Permission : GFDL, CC-by-sa.
Host factors
- Duration.
- Location.
- Inflammatory response.
- Genetics.
Mechanism
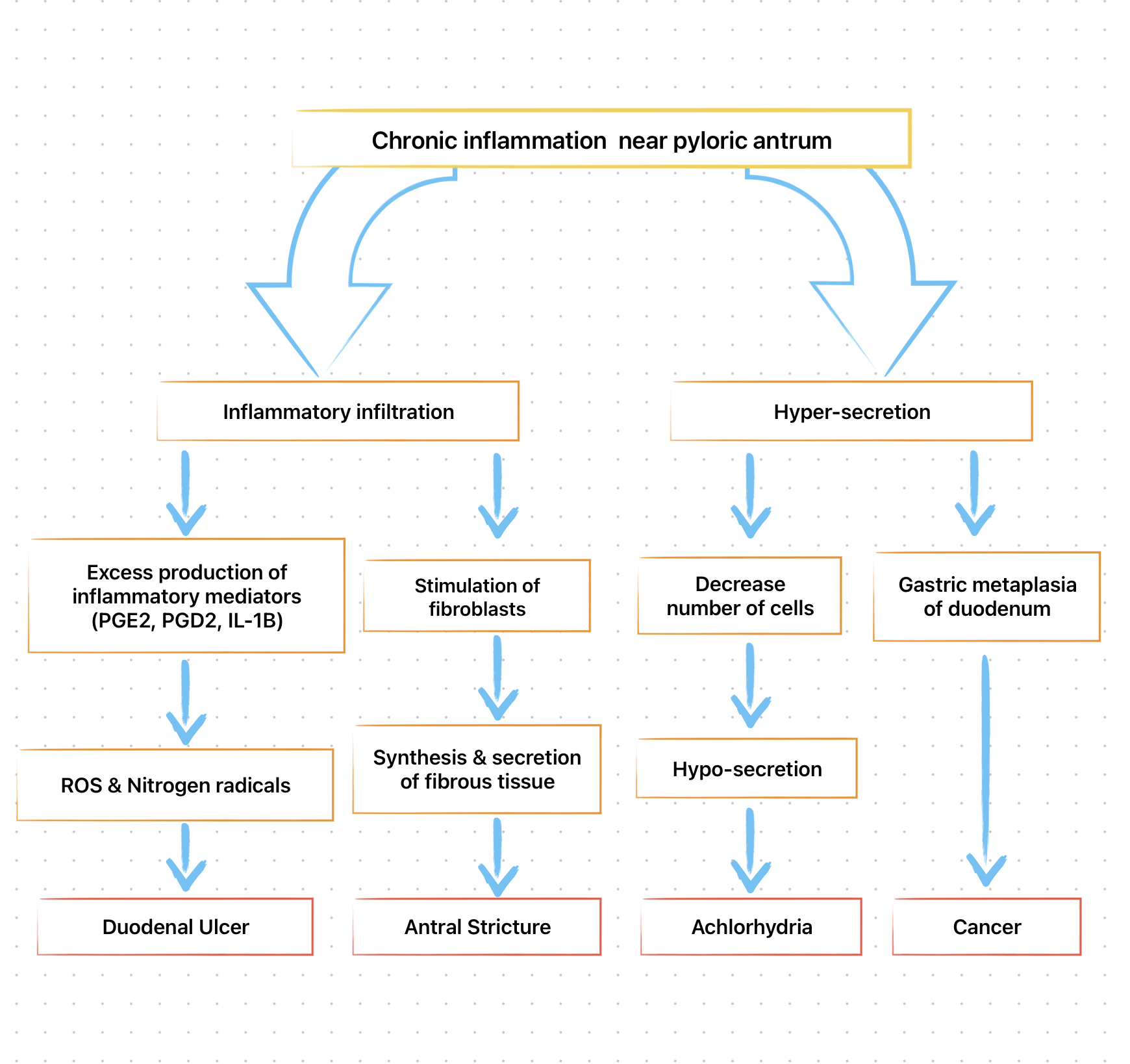
H. pylori is strategically designed to live within the aggressive environment of the stomach. It may attach to gastric epithelium, but under normal circumstances does not appear to invade cells.
-
H. pylori infection leads to an inflammatory response that includes recruitment of neutrophils, lymphocytes (T and B), macrophages and plasma cells.
-
Also, the bacteria causes local injury by binding to Class II MHC molecules expressed on gastric epithelial cells leading to cell death (apoptosis).
-
Bacterial strains that encode cag-PAI introduce Cag A into the host cells, leading to further cell injury and activation of cellular pathways involved in cytokine production and repression of tumor-suppressor genes.
-
It also leads to mucosal and systemic humoral response, which instead of eradication of the bacteria, further compounds cell injury.
-
Additional mechanisms by which H pylori may cause epithelial cell injury include :
- Activated neutrophil-mediated production of reactive oxygen or nitrogen species and enhanced epithelial cell turnover and
- Apoptosis related to interaction with T cells (TH1 cells) and interferon-y.
The type and distribution of gastritis correlate with the ultimate gastric and duodenal pathology observed.
Detection of H. pylori
-
Invasive tests (Endoscopy/ Biopsy required)
- Rapid urease.
- Histology.
- Culture.
-
Noninvasive tests
- Serology
- Urea breath test
- Stool antigen.
-
*Rapid urease and Urea breath test may show false negative results with recent use of PPIs, antibiotics or bismuth compounds.
Treatment for H. pylori infection
The goal is to provide relief of symptoms (pain or dyspepsia), promote ulcer healing and ultimately prevent ulcer recurrence and complications. Multiple drugs have been evaluated, however, no single agent is effective in eradicating the organism. Combination therapy for 14 days provides the greatest efficacy.
First-line therapy (Regimen)
- Clarithromycin triple : PPI (standard or double dose) + Clarithromycin (500mg) + Amoxicillin (1g) or Metronidazole (500mg TID) -> BID for 14 days.
- Bismuth quadruple : PPI (standard dose BID) + Bismuth subcitrate (120-300mg QID) or subsalicylate (300mg QID) + Tetracycline (500mg QID) + Metronidazole (250mg QID or 500mg TID to QID) for 10-14 days.
- Concomitant : PPI (standard dose) + Clarithromycin (500mg) + Amoxicillin (1g) + Nitroimidazole (500mg) -> BID for 10-14 days.
- Sequential : PPI (standard dose) BID for 5-7 days, followed by, PPI + Clarithromycin (500mg) + Nitroimidazole (500mg) BID for 5-7 days.
- Hybrid : PPI (standard dose) + Amoxicillin (1g) BID for 7 days, PPI, Amoxicillin, Clarithromycin (500mg), Nitroimidazole (500mg) BID for 7 days.
- Levofloxacin triple : PPI (standard or double dose BID) + Levofloxacin (500mg OD) + Amoxicillin (1g BID) for 5-7 days.
- Levofloxacin sequential : PPI (standard or double dose) + Amoxicillin (1g) BID for 5-7 days, followed by, PPI + Amoxicillin + Levofloxacin (500mg OD) + Nitroimidazole (500mg) BID for 5-7 days.
- LOAD : Levofloxacin (250mg OD) + PPI (double dose OD) + Nitazoxanide (500mg BID) + Doxycycline (100mg OD) for 7-10 days.
Salvage therapy
- Bismuth quadruple for 14 days.
- Levofloxacin triple for 14 days.
- Concomitant therapy for 10-14 days.
- Rifabutin triple : PPI (standard dose BID) + Rifabutin (300mg OD) + Amoxicillin (1g BD) for 10 days.
- High-dose dual regimen : PPI (standard or double dose TID or QID) + Amoxicillin (1g TID or 750 mg QID) for 14 days.
*Nitroimidazole includes Metronidazole and Tinidazole.
*FDA-approved regimen : Clarithromycin triple (PPI + Clarithromycin + Amoxicillin), and Bismuth quadruple (Bismuth subcitrate + Tetracycline + Metronidazole + PPI).
How to select antibiotics ?
-
Penicillin allergy : Negative; Macrolide exposure : No
- Bismuth quadruple.
- Concomitant.
- Clarithromycin triple with amoxicillin.
-
Penicillin allergy : Negative; Macrolide exposure : Yes (or in regions where clarithromycin resistance is known to be > 15%)
- Bismuth quadruple.
- Levofloxacin triple.
- Levofloxacin sequential.
-
Penicillin allergy : Positive; Macrolide exposure : No
- Clarithromycin triple with metronidazole.
- Bismuth quadruple.
-
Penicillin allergy : Positive; Macrolide exposure : Yes (or in regions where clarithromycin resistance is known to be > 15%)
- Bismuth quadruple.
Treatment Guidelines
The recommendations of Maastricht IV/Florence Consensus Report and American College of Gastroenterology (ACG) are as under:
-
Test-and-treat approach is recommended for,
- Patients with uninvestigated dyspepsia, if the local incidence of H. pylori is greater than 20%.
- Individuals greater than 60 years age with uninvestigated dyspepsia.
- Patients who will be using NSAIDs (including low-dose aspirin) on a long-term basis, especially if there is a prior history of PUD.
- Patients with unexplained iron deficiency anemia and idiopathic thrombocytopenic purpura.
-
Eradication of H. pylori indicated in,
- Patients who have undergone resection of early gastric cancer.
- First-degree relatives of family members with gastric cancer.
- Patients with previous gastric neoplasm treated by endoscopic or subtotal resection.
- Individuals with a risk of gastritis (severe pangastritis or body-predominant gastritis) or severe atrophy.
- Patients with gastric acid inhibition for more than 1 year.
- Individuals with strong environmental risk factors for gastric cancer (heavy smoking; high exposure to dust, coal, quartz, or cement; and/or work in quarries).
- H. pylori positive patients with a fear of gastric cancer.
Side effects of H. pylori eradication therapy
- Bismuth : May cause black stools, constipation, or darkening of tongue.
- Amoxicillin : Pseudomembranous colitis (1-2%), Antibiotic-associated diarrhoea, nausea, vomiting, skin rash, and allergic reaction. (May ameliorate with concomitant use of probiotics.)
- Metronidazole : Metallic taste (common), peripheral neuropathy (rare), flushing and vomiting when taken with food.
- Abdominal cramp.
- Potential for development of antibiotic-resistant strains.
Points to Note
-
The aim for initial eradication rates should be 85-90%.
-
Dual therapy (PPI + Amoxicillin, PPI + Clarithromycin, Ranitidine bismuth citrate + Clarithromycin) is not recommended as the eradication rates are less than 80-85%.
-
Poor patient compliance seen with triple drug therapy can be addressed by simplifying the regimens to twice a day.
-
Most common cause for treatment failure in compliant patients is the presence of an antibiotic resistant strain.
- The patient should be asked about prior antibiotic exposure and and the information should be considered for potential antibiotic resistance.
- Clarithromycin use should be excluded in patients with prior macrolide usage.
-
If eradication is not achieved in a compliant patient with both first-line and salvage therapy, culture and sensitivity should be considered.
-
If recurrent infection occurs within first 6 months after completing therapy, the most likely explanation is recrudescence as opposed to reinfection.
References
- Harrison's Principles of Internal Medicine (21st edition), Joseph Loscalzo & Anthony S. Fauci & Dennis L. Kasper & Stephen Hauser & Dan Longo & J. Larry Jameson, The McGraw-Hill Companies.
*This article is an excerpt from the above mentioned book and Medical Sutras does not make any ownership or affiliation claims.