Defluoridation can be defined as the downward adjustment of fluoride level in drinking water to optimal level of 1 ppm.
It involves removing excess, naturally occuring fluorides from drinking water, in order to improve the quality of water.
In India, the states having high fluoride levels (endemic fluoride belts with fluoride content in water more than 4.00 ppm) are:
- Punjab
- Haryana
- Rajasthan
- Gujarat
- Madhya Pradesh
- Andhra Pradesh
- Telangana
Characteristics
The defluoridation process should be:
- Cost-effective and efficient.
- Simple to use i.e., easy to handle or operate, with readily available raw materials.
- Consistent in performance over a long period.
- Acceptable to the community.
- Independent of input factors such as fluoride concentration, alkalinity, pH, temperature.
- Free from addition of undesirable substances such as Aluminium to the treated water.
- Have no adverse effect on taste, odor, and pH of water.
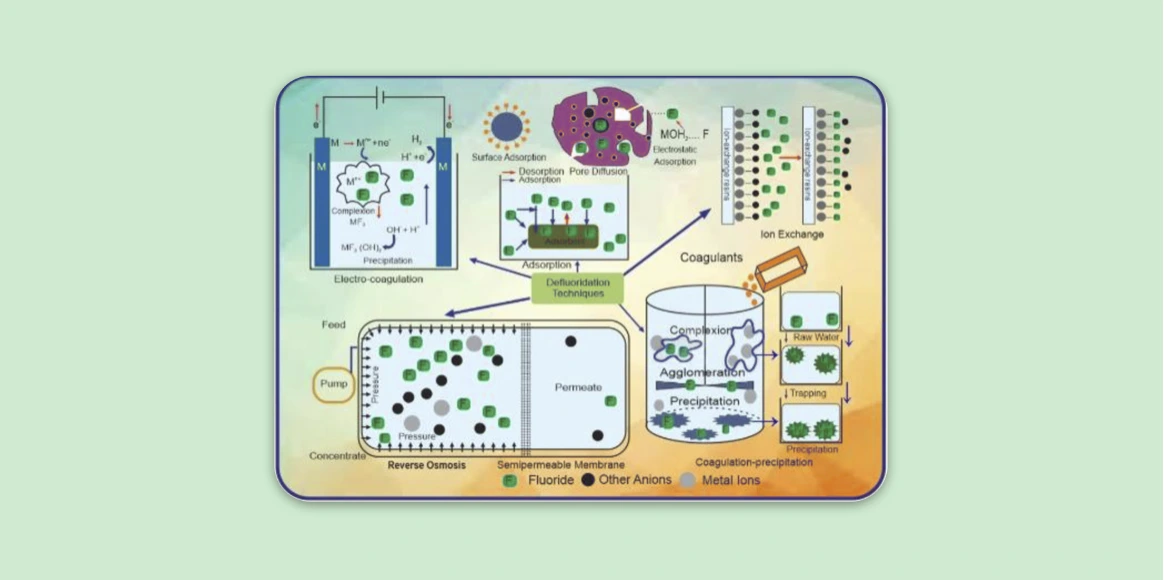
Techniques/Methods
Defluoridation can be achieved using following methods:
- Adsorption technique
- Ion-exchange technique
- Precipitation technique
- Other methods such as reverse osmosis and electrolysis.
Adsorption Technique
- It involves adsorption of fluoride ions onto the surface of an active agent, thus decreasing the concentration of fluoride in water.
- The agents include activated alumina, activated carbon, and bone char. Some of the natural adsorbents include seeds of the drumstick tree, roots of vetiver grass, and tamarind seeds.
- This process depends on suitable conditions like pH, temperature, flow rate and grain size of the material.
Ion-exchange Technique
It involves use of anion and cation exchange resins.
Anion exchange resins
- These include polystyrene anion exchange resins and basic quaternary ammonium type resins.
- Some disadvantages include fluoride removal on prolonged use, cost and alteration of water taste.
Cation exchange resins
- These include defluoron-1 (combination of sulphonated saw dust impregnated with 2% alum solution), carbion, magnesia, and defluoron-2 (sulphonated coal, works on the aluminum cycles).
Precipitation Technique
- These methods are based on the addition of chemicals (coagulants and coagulant aids), and subsequent precipitation of soluble fluoride salt as insoluble fluorapatite.
- Frequently used materials include aluminium salts (e.g. alum), lime, polyaluminum chloride (PAC), polyaluminum hydroxyl sulfate, and brushite.
Other Methods
- Reverse Osmosis: In this method, water is forced through a semipermeable membrane using hydraulic pressure, resulting in leaving the salts behind. There is about 45-90% removal of fluoride, while the pH of water raises from 5.5 to 7.
- Defluoridation of electrolysis: It involves adsorption of fluoride with freshly precipitated aluminum hydroxide (generated by anodic dissolution of aluminum or its alloys in an electrochemical cell).
References
- Nikhil Marwah, Textbook of Pediatric Dentistry, 4th Edition, Jaypee Brothers Medical Publishers (P) Ltd.
- Shobha Tandon, Textbook of Pedodontics, Paras Medical Publisher.
- S. S. Hiremath - Textbook of Public Health Dentistry, Elsevier India (2016). https://amzn.to/49mh2jw
- Image Source: https://pubs.rsc.org/en/content/articlehtml/2022/va/d1va00039j
*This article is an excerpt from the above mentioned sources and Medical Sutras does not make any ownership and affiliation claims.