Celiac disease is a small intestinal enteropathy, characterised by autoantibodies to tissue transglutaminase, resulting in hypersensitive immune response towards gluten and related proteins.
Synonyms: Celiac sprue, Non‐tropical sprue, Idiopathic sprue, Idiopathic steatorrhoea, Gluten-sensitive enteropathy.
Gluten: Found in foods produced from barley, rye, some varieties of oats and wheat (Mnemonic: BROW). It is a common additive to prepared foods and pharmaceuticals.
Epidemiology
- Global prevalence: 1.4%.
- Prevalence in first-degree relatives: 10-15%.
Etiology
Celiac disease is widely regarded as an autoimmune disease that occurs in genetically predisposed people.
Histocompatibility locus antigens HLA DQ2 and DQ8 are found in 25-30% of general population.
- Presence of one of the two haplotypes is necessary but not sufficient for developing celiac disease.
- Detection of these alleles is not useful for diagnosis, since, most carriers never develop celiac disease.
- Negative predictive value: More than 99%, very useful in ruling out celiac disease.
*Tissue transglutaminase deamidates glutamine residues of gluten-derived peptides, and facilitates their presentation by antigen-presenting cells.
Clinical Manifestations
Ranges from being asymptomatic to having isolated iron-deficiency anemia, and malabsorption of multiple nutrients.
- Common presenting complaints: Diarrhea, weight loss, and growth failure in children.
- Bloating and irregular bowel habits.
- Migraine headaches and ataxia.
- Osteoporosis, iron-deficiency anemia, and abnormal liver enzymes.
Associated Diseases
- Autoimmune disorders such as type 1 diabetes mellitus and autoimmune thyroid disease.
- Dermatitis herpetiformis, characterised by by a vesicular rash mediated by IgA deposits in the skin.
- Down syndrome and Turner syndrome -> Have an increased risk of celiac disease.
Mechanism of Diarrhea
- Villus atrophy in proximal small intestine, leads to steatorrhea from mucosal malabsorption.
- In addition, there is secretory component due to crypt hyperplasia and fluid hypersecretion from the crypt epithelium.
Diagnosis
- Initial Screening: Serum antibodies incl. tissue transglutaminase IgA, anti-endomysial and deamidated anti-gliadin antibodies.
- Serum IgA levels: To detect false-negative results that may occur due to IgA deficiency. Tissue transglutaminase IgG antibodies or deamidated antigliadin IgG antibodies are detectable and diagnostic in IgA-deficient patients.
- Confirmatory test: Endoscopy with small-intestinal biopsy.
- Family members of patients with celiac disease are screened if symptomatic.
Biopsy Findings
- Mucosal architectural changes: Villus atrophy, crypt hyperplasia, thickening of basement membrane under surface epithelium, and reduced number of goblet cells.
- Mucosal inflammation, incl. increased intraepithelial lymphocytes.
- Enterocyte changes: Cuboidal morphology, loss of basal nuclear orientation and cytoplasmic vacuoles.
Marsh Classification
The modified Marsh-Oberhuber classification categorises different types of celiac disease-related lesions and is used to quantify severity of disease involvement.
| **Marsh 3** | ||||||
|---|---|---|---|---|---|---|
| **Marsh 0** | **Marsh 1** | **Marsh 2** | **3A** | **3B** | **3C** | |
| **IEL**\* | < 30/100 | >30/100 | >30/100 | >30/100 | >30/100 | >30/100 |
| **Crypt hyperplasia** | - | - | + | + | + | + |
| **Villous atrophy** | - | - | - | Mild | Moderate | Total |
| Pre-infiltrative | Infiltrative | Infiltrative-hyperplastic | Flat destructive | |||
| \***IEL (Intraepithelial Lymphocytes)**: Number of intraepithelial lymphocytes per 100 enterocytes.
+Marsh 4 (For historic purpose): Atrophic-hypoplastic, IEL <30/100, Total villous atrophy. | ||||||
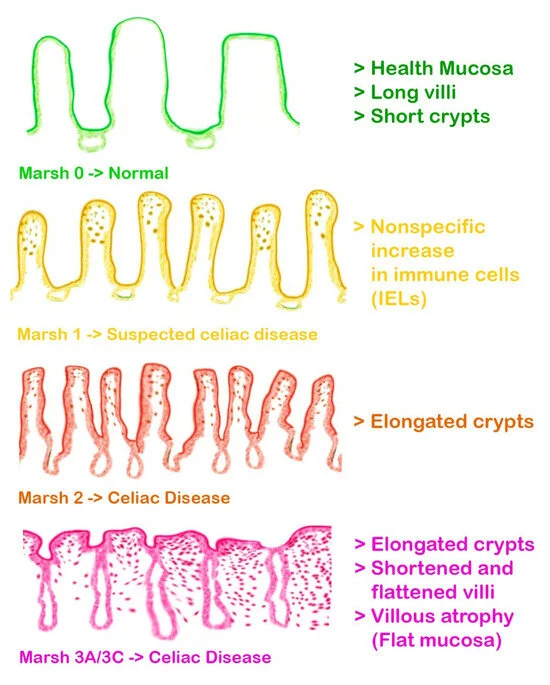
Image Source: https://www.mdpi.com/2077-0383/13/5/1382
Complications
- Refractory celiac disease.
- Enteropathy-associated T-cell lymphoma.
- Hyposplenism.
- Small-bowel adenocarcinoma.
Refractory Celiac Disease
- Found in about 10% of patients with persistent active disease.
- Characterised by ongoing diarrhea, weight loss and persistent villus atrophy on biopsy after 1 year of strict gluten-free diet.
- Also, the patients have negative celiac serology confirming adherence to gluten-free diet.
- Type 1 refractory celiac disease: Normal intraepithelial lymphocyte population.
- Type 2 refractory celiac disease: Clonal expansion of CD3+ intraepithelial lymphocytes that also contain a monoclonal rearrangement of the gamma chain of T-cell receptor. It has a worse prognosis, due to its association with T-cell lymphoma.
Treatment
The mainstay of treatment is institution of a strict gluten-free diet. Patients must be given rigorous dietary instruction from a dietician and adhere lifelong to a gluten-free diet.
Patients whose symptoms resolve after gluten-free diet
- Serologic follow-up to confirm compliance with the diet.
- Follow-up biopsy to document complete healing of villous atrophy.
Patient whose symptoms does not resolve after gluten-free diet
- The most common cause is nonadherence to diet or inadvertent gluten exposure.
- Biopsy, to determine the degree of disease activity and to rule out other causes and complications.
- If biopsy is negative, but, symptoms persist, other causes of abdominal pain and diarrhea associated with celiac disease are considered such as IBS, microscopic colitis, small-bowel bacterial overgrowth, and lactose/fructose intolerance.
References
- Harrison's Principles of Internal Medicine (21st edition), Joseph Loscalzo & Anthony S. Fauci & Dennis L. Kasper & Stephen Hauser & Dan Longo & J. Larry Jameson, The McGraw-Hill Companies.
- Coeliac disease: an update for pathologists B C Dickson, C J Streutker, and R Chetty J Clin Pathol. 2006 Oct; 59(10): 1008–1016, doi: 10.1136/jcp.2005.035345 PMCID: PMC1861744 PMID: 17021129
- The image used as cover photo is licensed under the Creative Commons Attribution-Share Alike 4.0 International license. Source: Wikimedia Commons. Author: www.scientificanimations.com
*This article is an excerpt from the above mentioned book and Medical Sutras does not make any ownership or affiliation claims.