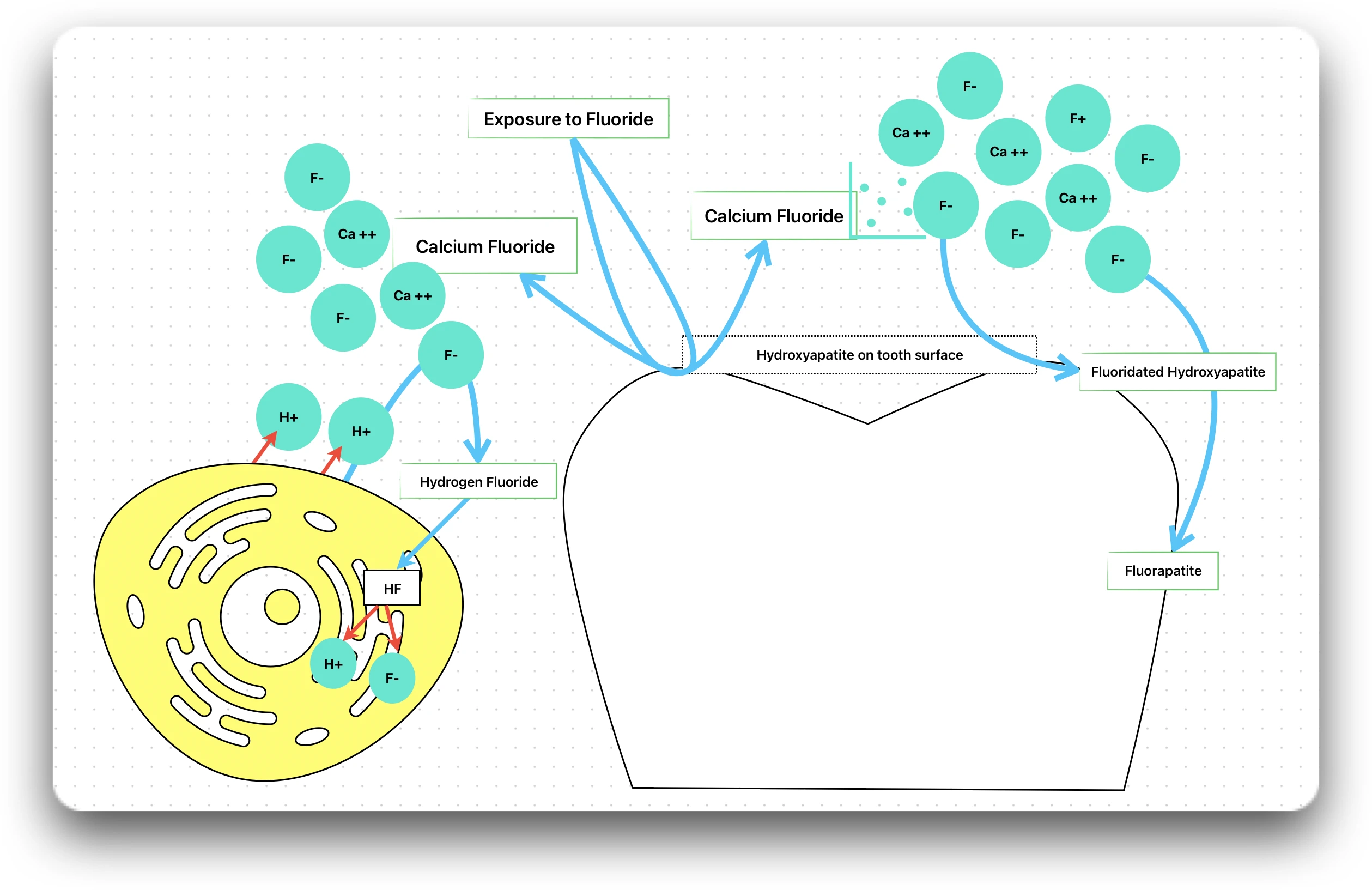
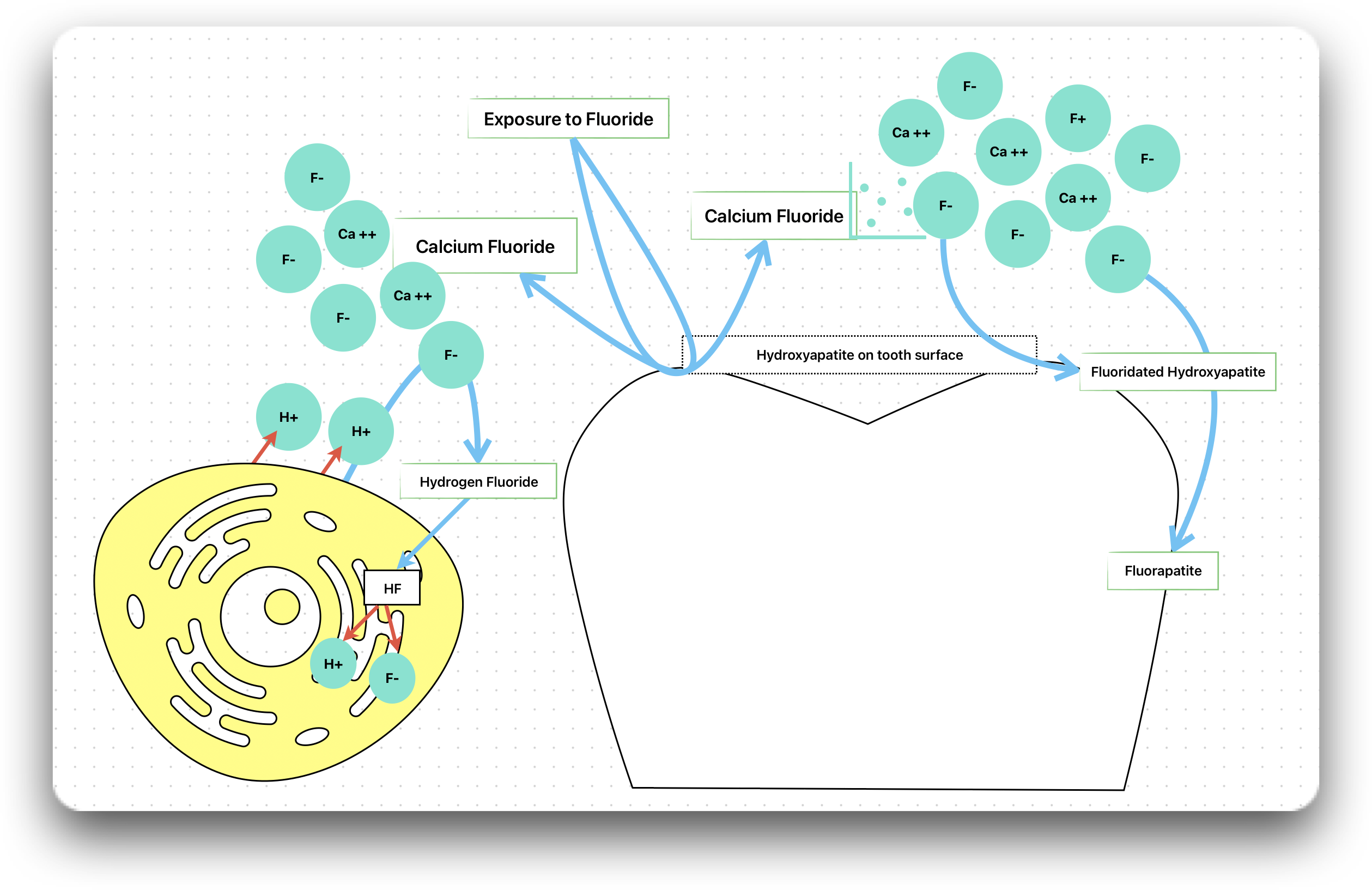
The cariostatic effect of fluorides is multifactorial, and the mechanisms by which fluorides increase caries resistance may arise from both systemic and topical applications of fluoride.
The proposed mechanisms that are assumed to work together include,
- Increased enamel resistance and reduction in solubility.
- Enhanced remineralisation of incipient lesions.
- Anti-bacterial action.
- Interference with plaque formation.
- Modification in tooth morphology.
It is claimed that formation of calcium fluoride (CaF2) in plaque and on the enamel surface is the most important factor in imparting the anti-caries effect.
Increased Enamel Resistance & Reduction in Solubility
Fluorides increase enamel resistance and reduce the solubility of enamel by,
- Incorporation of fluoride ions into the voids present in hydroxyapatite crystals (Void theory).
- Improved crystallinity.
- Formation of lesser acid soluble crystals with lower carbonate content.
- Enhancing the precipitation of hydroxyapatite and phosphate mineral, while preventing the formation of more soluble calcium phosphate, fluoride further reduces the solubility of enamel.
- Adsorption of the fluoride ions to the surface apatite crystals : When the bacteria generate acid, these ions will travel with the acid into the subsurface of the tooth, where they will adsorb to the crystal structure and protect it against being dissolved.
Void Theory
Normal tooth structure:
- The crystals in enamel rods are made up of hydroxyl, calcium and phosphate ions.
- The hydroxyl ions are present slightly above or below the plane formed by calcium ions and in order to maintain symmetry, equal number of hydroxyl ions must be present on both sides of the calcium plane.
- However, when the hydrogen of adjacent hydroxyl groups point towards each other, this results into stearic interference.
- Hence, to avoid this steric interference, one of the hydroxyl ions gets exterminated creating a void or reversal points.
- The presence of such voids in crystal structure decreases stability, leading to greater chemical reactivity and increase in solubility.
Fluoride incorporates into the voids to form fluorapatite
-
Fluoride ions fill up these voids and replace missing hydroxyl ions, thereby stabilising the crystal structure and decreasing the reactivity resulting in lower solubility and greater resistance to dissolution in acids.
-
Fluoride ions also helps in increasing the stability of apatite crystals by forming hydrogen bonds with neighbouring hydroxyl ions.
Improved Crystallinity
- Presence of fluoride leads to conversion of amorphous calcium phosphate into crystalline hydroxyphosphate.
- This results in larger crystals with fewer imperfection that produces less strain in crystal lattice, hence, stabilising the lattice.
Lesser Acid Solubility (Lower carbonate content)
- During tooth development, the enamel is initially made up of carbonated apatite crystals (highly soluble in acid), which are later replaced by hydroxyapatite crystals (less acid soluble).
- The carbonate ions cause major disturbance in the regular array of ions in the crystal lattice, which makes the carbonate-rich, calcium deficient regions susceptible to attack by the acid hydrogen ions.
- When the enamel mineralises under the influence of fluoride, the hydroxyapatite crystals get converted into fluoridated hydroxyapatite and further to fluorapatite (least soluble).
- In fluorapatite, the fluoride ions completely replace the hydroxyl ions of pure hydroxyapatite, making it highly resistant to dissolution by acid.
Enhanced Remineralisation of Incipient Lesions
- Destruction of enamel by caries is a cyclic phenomenon that consists of phases of demineralisation (dissolution of enamel and release of calcium, phosphate and other ions, when pH is less than 5.5) and re-mineralisation (precipitation of dissolved material when the bacteria stops producing acid).
- Fluoride enhances remineralisation by adsorbing to the crystal surface and attracting calcium ions, followed by phosphate ions, leading to new mineral formation.
- It speeds up the growth of new surface on the partially demineralised subsurface crystals. The new crystal surface layer is fluorapatite-like that have much lower solubility than the original carbonated hydroxyapatite crystals.
Antibacterial Action
As the bacteria produces acid, the pH of the plaque decreases and some of the fluoride ions present in the plaque fluid combines with hydrogen ions to form hydrogen fluoride.
Hydrogen fluoride (HF) is an undissociated weak acid that rapidly penetrates the bacterial cell membrane. Once inside the cell, the HF dissociates back into hydrogen and fluoride ions into the alkaline cell cytoplasm.
The fluoride ions interferes with the bacterial enzymatic activity, while the hydrogen ions acidifies the cytoplasmic compartment.
-
Fluoride inhibits the bacterial growth and metabolism by acting on several bacterial enzymes such as enolase and bacterial phosphatase.
- Prevents entry of glucose into the bacterial cell.
- Inhibits glycolysis, the process by which bacteria metabolise carbohydrate and produce acid.
- Prevents conversion of glucose to glycogen (storehouse when glucose is not available).
- Inhibits the production of extracellular polysaccharides by cariogenic bacteria (necessary for adherence of plaque to enamel surface).
- Stannous fluoride inhibits bacterial metabolism by oxidising the thiol group present in bacteria.
- Fluoride inhibits the normal process of proton outflow, further decreasing the intracellular pH. This makes the environment less favorable for many of the essential enzymes required for cell growth.
-
These inhibitory effects are related to the hydrogen ion concentration of the plaque, wherein a decrease in pH, leads to more fluoride ion formation and uptake into cells, thus, resulting in greater inhibitory action.
Interference with Plaque Formation
Lowering free surface energy
- Incorporation of fluoride in enamel (e.g., after treatment with metallic solutions such as stannous fluoride), reduces the free surface energy.
- This decreases the wettability of enamel, leading to reduced deposition of pellicle and subsequent plaque formation.
Desorption of protein and bacteria
- Higher concentration of fluoride in topical therapy may be effective in desorbing proteins and bacteria, as it inhibits the binding of acidic protein to the calcium site on the hydroxyapatite crystals.
- This results in lesser bacterial colonisation and reduced plaque accumulation.
Modification in Tooth Morphology
If fluoride is ingested during tooth development, evidence suggests that a more caries resistant tooth is formed due to selected inhibition of ameloblasts, with features such as,
- Rounder cusps.
- More shallow fissure with abrupt and tight approximation.
Points to Note
- Post-eruptive maturation: Newly erupted teeth often have hypo-mineralised areas that are prone to dental caries. Post eruptive maturation involves deposition of minerals into hypo-mineralised areas. Fluoride increases the rate of mineralisation or post-eruptive maturation of these areas.
- Very large amounts of fluoride in calcium phosphate solutions may actually inhibit remineralisation, by formation of calcium fluoride, which prevents hydroxyapatite crystal growth.
- Effect of fluoride on bacterias is concentration dependent: At low concentration, it retards acid formation; at higher concentration, it can have an effect on growth and metabolism; and at very high concentration it is bactericidal.
References
- C. M. Marya - A Textbook of Public Health Dentistry, Jaypee Brothers Medical Publishers (P) Ltd (2011). https://amzn.to/3SHP36G
- S. S. Hiremath - Textbook of Public Health Dentistry, Elsevier India (2016). https://amzn.to/49mh2jw
- Nikhil Marwah - Textbook of Pediatric Dentistry, Jaypee Brothers Medical Publishers Pvt. Ltd. (2018) https://amzn.to/3T3quT9
- Joseph John - Textbook of Preventive and Community Dentistry, CBS Publishers and Distributors Pvt. Ltd. (2018). https://amzn.to/3T0cI3B
- Soben Peter - Essentials of Public Health Dentistry (Community Dentistry), Arya Medi Publishing House Pvt Ltd (5th edition).
*This article is an excerpt from the above mentioned books and Medical Sutras does not make any ownership and affiliation claims.